BioPharma
Navigating through the Galectin Therapeutics (NASDAQ: GALT) Triangle In Search of Therapies
Published
2 years agoon
By
Boe Rimes
Galectin Therapeutics (NASDAQ: GALT), Galecto Biotech (NASDAQ: GLTO), and BioXyTran Inc. (OTCMKTS: BIXT)—these three companies all currently have market capitalizations under $100 million in the field of galectin research, one of the most promising fields of science which is suffering from the biotech beatdown where over 80+ biotechs are trading below cash value. GALT would be considered a microcap, whereas GLTO and BIXT would be considered nanocap stocks, with market capitalizations near or under $50 million. These tiny valuations are in contrast to the companies’ developmental pipelines, each with therapies in mid-to-late stage development—phase 2 or phase 3 clinical studies. The market is simply ignoring galectin-focused biotechs in what could become a field of research worth tens of billions in the next few years.
There are just a handful of companies in this emerging field of galectin research. These three biotechs are developing galectin inhibitors for some of humanity’s most significant unmet medical needs, ranging from liver disease to cancer to COVID-19. Galectins, particularly galectin-3, are implicated in a wide range of diseases and hold significant therapeutic promise. A quick browse through Google will reveal that a search of virtually any chronic disease plus galectin-3 will get multiple hits of medical journal articles recommending galectin-3 as a biomarker of disease severity or as a potential treatment.
Galectin Science Getting the Cold Shoulder
It is hard to fathom that something with so much potential is being utterly ignored. This is precisely why buying a basket of these stocks could result in an exceptional return. Investors may want to get in on the ground floor of this up-and-coming technology, but which company should they pick or should they pick all three and weigh them differently? Each of these three companies has its strong points and weaknesses. There is, however, one that remains the best of the breed.
 Galecto Biotech (GLTO): Cash King
Galecto Biotech (GLTO): Cash King
Many investors in this market are looking for biotechs with lots of cash such that they are not subject to volatile markets and in need of cash—usually via equity raises. The company is trading well below its liquid assets, valued at $97 million (cash and marketable securities), and has multiple synthetic galectin inhibitor formulations in its pipeline. Their lead candidate, GB0139, is a promising inhaled galectin-3 inhibitor for idiopathic pulmonary fibrosis (IPF)
Galecto was also testing its galectin-3 inhibitor in COVID-19 acute respiratory distress syndrome, but they appear to have discontinued the program in light of effective early-stage therapies (antivirals) being developed and in some cases approved, like Pfizer’s (NYSE: PFE) Paxlovid, and Merck’s (NYSE: MRK) molnupiravir. It is also possible they saw what BioXyTran was accomplishing with its galectin inhibitor Prolectin-M in mild-moderate COVID-19 and decided they could not compete.
Galecto is also advancing another oral galectin-3 inhibitor, GB1211, in liver cirrhosis and cancer. The company is measuring outcomes for NASH cirrhosis, a projected $54 billion market with no current approved therapies and hardly any candidates for late-stage NASH (cirrhosis), which accounts for over 40% of the cost burden of NASH. However, their phase 2 trial is only 12 weeks long and will almost certainly not detect any significant efficacy signals with a small sample size and short duration, as many NASH trials need to be 12-18 months or longer, especially in cirrhosis, to detect outcomes as liver scarring can take a while to reverse and patients whose livers decompensate on placebo may only do so at a slow pace. Additionally, it is not known whether the drug is very tolerable yet (though it may be) as cirrhosis patients have delicate livers and need an extremely well-tolerated drug.
The company’s oncology indication is promising as the company is in a phase 2 trial and recently published some research showing GB0139 reverses galectin-3 induced resistance of PD-1 inhibitors like Keytruda, which occurs due to galectin blockade. Galectin’s and BioXyTran’s galectin inhibitors are also known to synergize with cancer treatments to enhance outcomes.
Galectin-3 is a key player in the tumor microenvironment and therefore oncology remains a very promising indication for Galecto.
Galectin Therapeutics (GALT): Leader In Development and Clinical Testing
Galectin Therapeutics’ key differentiator from Galecto is its use of a naturally derived, refined complex carbohydrate as a galectin inhibitor. These compounds are known to be well-tolerated and have much better half-lives than the small synthetic compounds developed by Galecto; for instance, Galectin’s lead drug, belapectin, has a 20-hour half-life in NASH cirrhosis patients, whereas Galecto’s GB0139 (formerly TD139) has an 8hr half-life (IPF patients). Additionally, belapectin may be uptaken by macrophages rather than purely metabolized, meaning its therapeutic effect can persist inside the main source of galectin-3, the macrophage. For this reason, the therapy is very targeted and well-tolerated, reportedly reducing side effects (rather than adding side effects) when used in combination with Keytruda (pembrolizumab) in cancer therapy. Keytruda is projected to be the best-selling drug on the planet.
Galectin Therapeutics is pursuing belapectin treatment for NASH cirrhosis, primarily, with a 3-year, robust global phase 2b/3 trial assessing the progression of cirrhosis patients developing esophageal varices, which occur due to significant liver pressure buildup from fibrosis. Their phase 2 study showed that patients on belapectin developed fewer varices than those on placebo, the only NASH cirrhosis clinical trial to show signs of clinical efficacy.
 The company has also conducted preliminary studies in cancer—advanced melanoma and head and neck squamous cell carcinoma—and has demonstrated likely synergy with PD-1 inhibitors like Keytruda with its phase 1 study and some preclinical studies. Notably, the therapy is expected to improve clinical outcomes and also potentially the side effect profile of PD-1 inhibitors. Checkpoint inhibitors are known to unfortunately induce autoimmune or inflammatory side effects in patients, causing them to have to pause or terminate therapy, which can further have a detrimental effect on their underlying condition. A therapy that can ameliorate the side effects and improve the efficacy of, for instance, Merck’s Keytruda, could be worth a lot to patients and Big Pharma. Galectin-3 expression predicts response to checkpoint inhibitor response in various tumors, notably non small cell lung cancer (NSCLC).
The company has also conducted preliminary studies in cancer—advanced melanoma and head and neck squamous cell carcinoma—and has demonstrated likely synergy with PD-1 inhibitors like Keytruda with its phase 1 study and some preclinical studies. Notably, the therapy is expected to improve clinical outcomes and also potentially the side effect profile of PD-1 inhibitors. Checkpoint inhibitors are known to unfortunately induce autoimmune or inflammatory side effects in patients, causing them to have to pause or terminate therapy, which can further have a detrimental effect on their underlying condition. A therapy that can ameliorate the side effects and improve the efficacy of, for instance, Merck’s Keytruda, could be worth a lot to patients and Big Pharma. Galectin-3 expression predicts response to checkpoint inhibitor response in various tumors, notably non small cell lung cancer (NSCLC).
The spider plot above shows how much of a clear difference galectin-3 may make for the patient’s response to therapy and the enormous potential galectin inhibitors have in improving immunotherapy, and this is a key piece of evidence for all three of the galectin companies. Galectin’s belapectin is extremely promising in oncology, making the entire company very attractive given its financial support from Chairman of the Board and billionaire Richard Uihlein, but the main drawback with the company is that it is slow, having taken about 2 years to design a phase 3 trial and about 5 years so far to move from phase 1 in oncology to a planned phase 2.
BioXyTran Inc. (BIXT): Best Indications and Formulations, Dark Horse
BioXyTran may not have the cash on hand that Galecto does, nor the number of patients dosed that Galectin has, but it may have potentially the best galectin inhibitor formulations and clinical indications to bring to market. The Company’s pipeline slide makes it difficult to ascertain where exactly they are in the clinical trial process, but their statistically significant finding in their pilot study was without a doubt an international breakthrough that was largely ignored or drowned out but the overwhelming focus on the vaccination effort.
In this peer-reviewed study, an oral chewable tablet called Prolectin-M eliminated viral load to undetectable levels in symptomatic COVID-19 patients within 3 days. In their upcoming PROTECT study, they are expected to enroll 408 patients “regardless of vaccine status, viral variant, or underlying medical conditions.” This enrollment criterion is noticeably different than every other clinical trial in COVID-19 which seeks to exclude healthy subjects that can get better on their own. They are choosing to go after the hardest-to-treat group which is for the most part healthy subjects that show symptoms of infection. On the surface, this type of trial design is risky, but there is a very subtle inclusion criterion that highlights the skill of BioXyTran’s regulatory team. Their strategy revolves around cherrypicking (selecting) patients that are symptomatic with high viral loads defined by a Ct value <25. Even a healthy patient with this high viral load is going to take well over a week to recover. If their statistical significance holds, then viral elimination should occur in 3 days versus a week or more. The BIXT team has to be extremely confident in their mechanism of action (MOA) to pursue what many pundits would call a risky trial design. Prolectin-M is shaping up to be potentially the most effective COVID-19 antiviral in existence.
The approval of Paxlovid and molnupiravir was based on reduction of the risk of hospitalization or death. Prolectin-M would likely need to demonstrate efficacy in the same endpoint. The rationale for Prolectin-M meeting its endpoints so quickly is based on an MOA that doesn’t need a competent immune system to eliminate the virus. The galectin antagonist binds to the spike protein and prevents viral entry and then eventually gets filtered out of the liver with the virus in tow.
 Recently, a real-world study was conducted with the approved Merck and Pfizer antivirals (Paxlovid and Molnupiravir), and the time to undetectable COVID-19 was measured. Paxlovid outperformed molnupiravir, but when Prolectin-M is plotted against Paxlovid and control, the night and day difference in efficacy can be observed. In 20 days of using Paxlovid, approximately 30% of the patient population reached an undetectable level of Ct value. In theory, 100% of the patient population would reach undetectable levels in 3 days using Prolectin-M (depicted with the black line).
Recently, a real-world study was conducted with the approved Merck and Pfizer antivirals (Paxlovid and Molnupiravir), and the time to undetectable COVID-19 was measured. Paxlovid outperformed molnupiravir, but when Prolectin-M is plotted against Paxlovid and control, the night and day difference in efficacy can be observed. In 20 days of using Paxlovid, approximately 30% of the patient population reached an undetectable level of Ct value. In theory, 100% of the patient population would reach undetectable levels in 3 days using Prolectin-M (depicted with the black line).
 Adapted from Real-world effectiveness of molnupiravir and nirmatrelvir/ritonavir among COVID-19 inpatients during Hong Kong’s Omicron BA.2 wave: an observational study
Adapted from Real-world effectiveness of molnupiravir and nirmatrelvir/ritonavir among COVID-19 inpatients during Hong Kong’s Omicron BA.2 wave: an observational study
Paxlovid is expected to do $22 billion in sales this year. The largest order of pharmaceuticals in history was the $5.3 billion order for 10 million doses of Paxlovid. If BIXT can show better safety and efficacy over Paxlovid, a new record could be set. One thing that Prolectin-M clearly outperforms on is safety. There are no safety signals and galectin inhibitors as a class of drugs are quite safe. On the other hand, Pfizer’s Paxlovid is paired with an HIV medicine called ritonavir in order to slow the metabolism of the actual antiviral compound (nirmatrelvir) in order to use less to get the same effect avoiding any safety signals, and to improve the pharmacokinetic profile of nirmatrelvir.
Though BioXyTran’s primary pipeline candidate for COVID-19 is promising, the company shows a number of additional indications in its pipeline, including cancer and NASH using galectin inhibitors. Finally, the company has an oxygenation platform that is highly promising for stroke and organ transplantation which acts as an oxygen transport molecule that can perform the same functions as hemoglobin on red blood cells without blood type matching, degradation, or getting stuck in clots.
Galectin Comparison
When it comes to drug design and the best science, BIXT has the newest galectin inhibitor that was developed within the last 2 years. Looking at their presentations, BIXT carefully stayed away from specifically calling their molecules galectin-3 inhibitors as their competitors do; instead, they called them galectin antagonists. BioXyTran’s galectin antagonists clearly target galectin-3, so there has to be more to the story or perhaps a trade secret. Galectins 1, 3, and 9 are modulators of viral infection. It is reasonable to think their antagonist is targeting multiple galectins as multiple galectins mediate different processes in disease pathology (specifically galectin-1, galectin-3, and galectin-9 in multiple viral infections). Belapectin has shown in structural studies that it inhibits galectin-1 and galectin-3, and Galecto’s drugs are only really galectin-3 inhibitors. Obviously, galectin-3 is the most important, but there could be benefits to antagonizing multiple galectins.
In terms of clinical trials, GALT seems to hold the lead. They have done clinical trials in liver fibrosis, NASH cirrhosis, atopic dermatitis, psoriasis, melanoma, and head and neck squamous cell carcinoma. They also have dosed many patients. GLTO is doing clinical trials in idiopathic pulmonary fibrosis (IPF), NSCLC, liver cirrhosis, and myelofibrosis. BIXT currently only has an active indication in acute COVID-19 but from their recent patent approvals, it seems they are about to announce something new. While GALT has a commanding lead, they could easily lose the race to commercialization once BIXT starts a phase 2 or phase 3 trial in COVID-19. The trials in COVID-19 represent a much shorter pathway to approval than a NASH cirrhosis study which can take a number of years.
In terms of cash, GLTO is the strongest of the bunch with $3.7/share in cash. GALT comes in second with $0.53/share in cash. However, GALT does have the support of its Chairman of the Board, Richard Uihlein, who has consistently supported the company with lines of credit as well as above-the-market priced convertible notes. BIXT comes in last with approximately $700,000 and it simply needs money to move forward; this is the company’s primary problem, which should be surmountable given its clinical trial results and assets.
Investment Summary
With close to 4,000 journal articles on galectins suggesting their use as a prognostic biomarker of disease or as a therapeutic target, it’s imperative that a galectin inhibitor reaches approval. The top 3 companies in the space each have their strengths but none has the complete package. GALT has been the undisputed leader for years, but their slow management and one-molecule development has left them vulnerable to being overtaken by their rivals. GLTO has the weakest technology platform with its small molecule architecture, but it was able to boost its pipeline to 4 drug candidates. Their cash horde is significant but management has little skin in the game and has only retained 9.6% cumulative ownership of the company. BIXT seemingly came out of nowhere and has one if not two molecules in the clinic and is zipping through clinical trials. They have massive insider ownership and the most robust technology platforms, but they have little-to-no cash to develop their product. Their management team is led by Dr. Platt, who is arguably the grandfather of galectin science and has invented the greatest number of carbohydrate drugs and commands the greatest clinical trial experience.
The most impressive molecule award goes to Prolectin-M, developed by BIXT. It eliminated the SARS-CoV-2 virus to undetectable levels in 3 days (p=.029). It has the quickest and cleanest pathway to approval and is simply waiting for a larger, short-duration confirmatory trial before commercialization which means there is very little execution and regulatory risk. There is a very real possibility that this drug could end the pandemic by dramatically slowing the transmission of the disease and stopping virtually all hospitalizations in treated patients. BIXT appears best in breed followed by GALT.
Disclosure: MicroCapDaily and its owners do not have a position in any of the stocks mentioned and have posted this article for free without editorial input. This article was written by a guest contributor and solely reflects his opinions.
Image by Michal Jarmoluk from Pixabay

You may like
-


Bioxytran (OTCMKTS: BIXT) Peer-Review Published Showing Functional Cure for COVID-19
-


BioXyTran Inc (OTCMKTS: BIXT) Crushes Primary Endpoint, Expected to Achieve Unicorn Status in New Year
-


Galectin Runners Bioxytran Inc (OTCMKTS: BIXT) & Galectin Therapeutics (NASDAQ: GALT) Breaking Out
-


The Steady Rise of Bioxytran Inc (OTCMKTS: BIXT)
BioPharma
Brainstorm Cell Therapeutics’ (NASDAQ: BCLI) Post-Setback Resurgence: Unpacking FDA Impact & Market Expectations
Published
8 months agoon
November 30, 2023
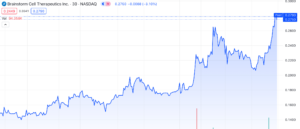
Brainstorm Cell Therapeutics (NASDAQ: BCLI) continues to gain momentum with a surge of over 125% since its low on October 31st, 2023. Following an unfavourable FDA decision on its ALS treatment, the stock hit an all-time low of $0.13 per share. Nethertheless, the stock experienced a significant rebound today, November 30th, 2023, rising by 26%, without accompanying press releases or SEC filings.
This resurgence prompts us to evaluate the possibilities for yet another company struggling to meet NASDAQ’s minimum bid requirements. A closer examination of recent months reveals intriguing developments that might pique the interest of potential investors. Our findings suggest BCLI holds promising near term prospects that could significantly alter its course.
Background:
BCLI specializes in developing therapies that utilize a patient’s own stem cells to combat severe neurodegenerative ailments like Amyotrophic Lateral Sclerosis (ALS) and progressive Multiple Sclerosis (MS). ALS affects nerve cells in the brain and spinal cord, while MS affects the central nervous system, causing problems with vision, balance, and muscle control.
The company holds exclusive global rights for the clinical advancement and commercialization of the NurOwn® technology platform, enabling the production of autologous MSC-NTF cells. These cells have received significant recognition and were awarded Orphan Drug designation by both the FDA and the European Medicines Agency (EMA).
Autologous MSC-NTF cells:
The way Autologous MSC-NTF cells work is very interesting. They’re a specific cell type obtained from an individual’s mesenchymal stem cells (MSCs). These cells are then genetically altered to produce neurotrophic factors (NTFs), which are designed to support and stimulate the growth and survival of nerve cells within the nervous system.
BrainStorm has conducted a Phase 3 clinical trial (NCT03280056) investigating the safety and effectiveness of multiple doses of autologous MSC-NTF cells for ALS. This trial received support from the California Institute for Regenerative Medicine, the ALS Association, and I AM ALS. Additionally, the company completed a Phase 2 open-label multicenter trial (NCT03799718) for progressive MS using these cells, backed by a grant from the National MS Society (NMSS).
Back to Square One:
Unfortunately for BCLI, 17 FDA advisory panel members voted that the data presented for their Phase 3 clinical trial for ALS did not demonstrate substantial evidence of the effectiveness of NurOwn for the treatment of mild-to-moderate ALS. There was simply only one member who voted in favour and another member that abstained from voting entirely.
Predictably, this resulted in a substantial downturn in the company’s share price, causing BCLI to plummet 87% below NASDAQ’s minimum bid compliance threshold of $1.00.
Subscribe to Microcapdaily.com Right Now by entering your Email in the box below.
Subscribe to Our 100% Free Penny Stock Newsletter. We Have Something Big Coming!
Is it Over?:
To those not well-versed in this matter, it might appear as if BCLI is forced to throw in the towel and start everything from scratch, but the reality is actually quite different.
When a drug fails in clinical trials, the pharmaceutical company or researchers typically reassess the data to understand why the drug failed.
They might conduct further analysis to determine if there were unforeseen side effects, issues with the study design, or if the drug simply didn’t show the desired effectiveness. They’ll work to make adjustments, refine the drug’s formulation, or in some cases, explore other alternatives by re-designing new clinical trials to evaluate the drug’s safety & efficacy for a different condition.
If none of those efforts yield a positive outcome, it’s typical to halt further development of that specific drug.
Regardless of the FDA’s decision, Co-CEO Lindborg mentioned, “We firmly believe that the data for NurOwn presented today provide a compelling case for approval, with clinical evidence in those with less advanced disease supported by strong and consistent biomarker data that are predictive of clinical response,”
Unfortunately for Lindborg, the decision does not rest with her. A panelist from the FDA, Lisa Lee, expressed, “Providing false hope can be ethically problematic and false hope is provided when the probability of a positive outcome is overestimated. And I think that seems to be the case here.”
After the devastating press release on September 27th, 2023, most of the retail crowd was on board with the FDA, mentioning it was time for BCLI to pack up, and go home – especially after seeing these comments from the FDA.
What Happened:
Amid FDA skepticism and investor uncertainties, BCLI took decisive action, securing a chance for a new phase 3 trial to showcase the effectiveness and safety of their NurOwn® technology for ALS treatment.
On October 24th, 2023, BCLI issued a press release affirming their conviction in the compelling data and expressing their intent to further substantiate it to the FDA.
More specifically, this was a “Strategic realignment” designed to:
- Support the company plans to conduct a double-blind, placebo-controlled Phase 3b U.S. clinical trial for NurOwn in ALS with an open-label extension and
- Continue to publish data from NurOwn’s Phase 3 clinical trial on biomarkers, long-term safety, survival, and the Expanded Access Program, providing transparency around NurOwn data and progressing ALS drug development.
Big Release:
Then on November 20th, 2023, BCLI issued another press release, stating that the FDA granted them an in person meeting to discuss the regulatory path forward for NurOwn® in ALS.
Within’ the last month or so, it appears the market is HIGHLY anticipating the outcome of this meeting, given it is now scheduled to take place on December 6, 2023.
This meeting allows BCLI to have a Special Protocol Assessment (SPA) with the FDA to agree on the overall protocol design for a confirmatory Phase 3 trial in ALS.
If you aren’t familiar with a SPA, it’s an agreement between a pharmaceutical company and the FDA regarding the design, endpoints, and size of a clinical trial intended to support a New Drug Application (NDA) or Biologics License Application (BLA). This agreement aims to confirm that the trial design, data analysis, and endpoints are acceptable to the FDA for the regulatory approval process.
A favorable result from this meeting could greatly benefit BCLI, paving the way for the actual launch of NurOwn®. This outcome stands in stark contrast to the prior belief, post the September 27th, 2023 news, which led many to assume otherwise.
If the trend remains positive, BCLI could see a significant surge in valuation, given the substantially reduced risk associated with a drug nearing commercial availability.
Conclusion:
Boosting the optimism for December 6th is the heightened enthusiasm among retail investors surrounding the company. Some speculate that the surge might mirror what SNGX experienced today, on November 20th, 2023. With technical day traders and swing traders in the mix, many perceive this as the final opportunity to join the trend before it gains further momentum. It’s crucial to remain vigilant as December 6 approaches rapidly, potentially ushering BCLI into an entirely altered and positive scenario.
We will update you on BCLI when more details emerge, subscribe to Microcapdaily to follow along!
Subscribe to Our 100% Free Penny Stock Newsletter. We Have Something Big Coming!
Disclosure: We have not been compensated for this article/video. MicroCap Daily is not an investment advisor; this article/video does not provide investment advice. Always do your research, make your own investment decisions, or consult with your nearest financial advisor. This article/video is not a solicitation or recommendation to buy, sell, or hold securities. This article/video is our opinion, is meant for informational and educational purposes only, and does not provide investment advice. Past performance is not indicative of future performance.
Picture by ckstockphoto from Pixabay.com
BioPharma
Apollomics (NASDAQ: APLM) Market Momentum: Pipeline Potential, Market Trends, and Retail Insights
Published
8 months agoon
November 27, 2023
Apollomics, Inc. (NASDAQ: APLM) has seen a significant surge in its shares, rising by ~20% at time of writing, accumulating a staggering 58% gain since November 21st, 2023. Surprisingly, there haven’t been any noticeable press releases or filings to explain this sudden valuation shift, so it’s prompted a closer examination on our end. Fortunately enough, we found the force driving the substantial gain, but first let’s look into APLM’s background to build a better picture of the opportunity at hand.
 Background:
Background:
APLM’s emergence as a public company is relatively recent, stemming from its merger with Maxpro Capital Acquisition Corp. (Nasdaq: JMAC) on March 30th, 2023. This merger also involved a $23.65 million private investment in public equity (PIPE) financing, a typical move when companies go public, aiming to secure capital. With this recent funding, APLM anticipates financial support through mid-2024, empowering the company to push forward with its ambitious lineup of 9 drug candidates.
APLM stands as an innovative player in the field of clinical-stage biopharmaceuticals, specializing in crafting cancer therapies that work hand-in-hand with the immune system and target precise molecular pathways to combat cancer. With a focus on oncology, the company boasts a lineup of nine potential drug candidates, six of which are actively progressing through clinical development stages.
Among its flagship projects are vebreltinib (APL-101), a potent c-MET inhibitor that targets multiple forms of non-small cell lung cancer (NSCLC), and uproleselan (APL-106), an E-Selectin antagonist designed to potentially enhance standard chemotherapy in treating acute myeloid leukemia.
Latest Release:
APLM recently announced significant news concerning its Chinese partner, Avistone Biotechnology, on November 11th, 2023. Avistone received conditional approval from China’s National Medical Products Administration (NMPA) to market vebreltinib. This medication is intended for treating a particular type of lung cancer known as MET exon 14 skipping non-small cell lung cancer (NSCLC).
Dr. Guo-Liang Yu, Apollomics’ leader, praised Avistone for this achievement, saying it’s a big step toward finding better treatments for tough cancers. Vebreltinib is a powerful medicine that targets how cancer cells grow. It could be a breakthrough for people with this specific lung cancer and other cancers caused by similar changes in cells.
Apollomics is talking with the U.S. Food and Drug Administration (FDA) about using vebreltinib to treat this kind of lung cancer in the U.S. They’re using data from their global study and Avistone’s study in China to do this.
It’s no secret, Lung cancer is a big problem, especially for people with non-small cell lung cancer (NSCLC), which is responsible for most lung cancer cases. This specific mutation has a significant unmet need, and APLM’s vebreltinib could be hope for those battling the devastating disease.
Subscribe to Microcapdaily.com Right Now by entering your Email in the box below.
Subscribe to Our 100% Free Penny Stock Newsletter. We Have Something Big Coming!
Market Opportunity:
To keep things simple, we’ll continue focusing on APLM’s Vebreltinib (APL-101), which is just 1 of 9 drugs in their pipeline.
This specific drug, cultivated through a collaboration with Beijing Pearl Biotechnology (now merged with Avistone Biotechnology), appears to display considerable potential. Its appeal lies in its demonstrated safety, effectiveness, and its potential to address a specific market segment where there’s a significant demand for treatments due to unmet medical needs.
Vebreltinib (APL-101):
APL-101 is being evaluated for its potential use in combating specific types of Non-Small Cell Lung Cancer (NSCLC), and a particularly aggressive form of brain cancer, secondary glioblastoma multiforme (sGBM).
APL-101 is a Type 1b class highly selective c-MET inhibitor. More simply put, it’s a drug that effectively targets and blocks the c-MET protein.
c-MET inhibitors have a wide potential range of markets due to the involvement of the c-MET pathway in various cancers and other diseases. The c-MET receptor, also known as the hepatocyte growth factor receptor (HGFR), plays a crucial role in cell growth, survival, and migration. Dysregulation of the c-MET pathway is associated with tumor growth, metastasis, and resistance to certain therapies in multiple cancer types.
Outside of NSCLC and sGBM, Apollomics has already seen promising effects inhibiting tumors related to certain types of cancers like gastric, hepatic, and pancreatic. But to keep things simple, we’ll stick to Apollomics core focus in NSCLC and sGBM.
The Market:
According to statistics published by the World Health Organization (WHO) from February 2022, 2.2 million patients were diagnosed with lung cancer in 2020, and non-small cell lung cancer accounted for nearly 85% of all lung cancer patients.
The global non-small cell lung carcinoma (NSCLC) market is expected to garner a market value of US$ 8.25 Billion in 2023 and is expected to accumulate a market value of US$ 21.40 Billion by registering a CAGR of 10% in the forecast period 2023 to 2033.
Delving deeper into particular types of NSCLC, the market potential for Exon-14 skip mutated NSCLC, c-MET amplifications in NSCLC (denovo), and c-MET amplifications in NSCLC (resistance driven) collectively represents a 3 billion-dollar opportunity.
In contrast, the market size for the population affected by epidermal growth factor receptor (EGFR) mutated NSCLC stands at a larger 7 billion-dollar opportunity.
Subscribe to Microcapdaily.com Right Now by entering your Email in the box below.
Subscribe to Our 100% Free Penny Stock Newsletter. We Have Something Big Coming!
SPARTA Trial:
SPARTA is part of a larger study combining Phase 1 and Phase 2 trials, conducted in 13 countries, 90+ sites and 370+ patients worldwide. It aims to understand how safe and effective vebreltinib is in treating various cancers. More specifically, it’s looking at non-small cell lung cancer (NSCLC) with a specific genetic mutation called c-MET exon 14 skipping, as well as other cancers with c-MET amplification or fusions.
The trial involves different groups of patients: those new to treatments targeting the c-MET protein and those who have previously received such treatments for NSCLC with the mutation. Additionally, it includes patients with various solid tumors showing c-MET amplifications or fusions, including glioblastoma multiforme, a severe type of brain cancer.
The main goal of SPARTA is to measure how well vebreltinib works by assessing the overall response rate (ORR) and how long the response lasts (duration of response) using specific evaluation criteria for each type of tumor. Secondary goals involve looking at any side effects and additional measures like time to progression, progression-free survival, and overall survival.
Overall the clinical findings show that vebreltinib could be a promising new treatment for cancers caused by MET alterations, especially in Non-Small Cell Lung Cancer (NSCLC) with MetEx14 skipping mutation. In a phase 2 trial, there was 75% overall response rate in patients with advanced NSCLC.
If you’d like more formal clinical trial information for vebreltinib, it’s is available on clinicaltrials.gov under the Phase 1/2 SPARTA trial (NCT03175224).
Big Picture:
This is just 1 of 9 drugs in APLM’s pipeline, and APL-101 is just 1 of 2 key assets. With our initial examination of the company, we were taken aback by the comparatively low valuation despite the promising pipeline. Like many, there has been a considerable decline in share value since their public listing.
Now that China’s National Medical Products Administration (NMPA) has given conditional approval, the company intends to get Vebreltinib (APL-101) approved in the U.S. and has met with the FDA in July. With an existing approval by the NMPA, receiving the NDA Approval from the FDA should realistically be a much quicker process.
On top of this, more interesting developments have occurred from online sources, indicating substantial short activity, further hinting at the possibility of APLM being the next big short squeeze candidate.
What Happened & Retail Insights:
APLM seems to be gaining attention across various social media platforms, notibly Twitter and Reddit.
Numerous investors are discussing on different platforms that APLM might become a potential candidate for a short squeeze. Users have pointed out that the cost to borrow shares has risen significantly. This increased short activity seems to have also led to a shortage of available shares to borrow.
Increased buying activity is likely to intensify the pressure on short sellers, compelling them to buy back shares to cover their positions. Given today’s substantial surge in trading volume, short sellers may find themselves in a tight spot, potentially regretting their initial choice to wager against APLM.
We came across additional analysis (due diligence) about APLM on Reddit, providing concise insights. This is but one piece of content that’s drawing increased interest in the stock among retail investors.
APLM – Apollomics Inc- DD THREAD – BIO STOCK –
byu/StockProfilers inpennystocks
In recent times, Reddit has become a hub for discussions regarding stocks and potential market movements. It gained particular prominence in 2020 as a platform for detailed research and discussions about stocks, witnessed notably during the retail trading frenzy involving stocks like GME (GameStop) and AMC (AMC Entertainment Holdings).
Analyst Ratings:
Two analysts have set notably optimistic price targets for APLM, suggesting it might reach $21.00, representing a potential 1650% increase from its current value. Additional sources offer different projections, with estimates of $11.00 (an 816% increase), $14.00 (a 1066% gain), and $17.00 (a 1326% rise). It’s crucial to understand that analyst ratings are not certainties but rather projections based on their research. We commonly advise examining past coverage to assess the accuracy of analysts’ ratings and forecasts.
Conclusion:
In summary, APLM experienced a substantial surge, seemingly fueled by retail investors countering short sellers. However, beyond the retail-driven buzz speculating a potential short squeeze akin to TPST, a deeper narrative unfolds, signalling a compelling long-term proposition.
APLM’s pipeline exhibits fairly de-risked assets and promising data across multiple indications, not to mention large amount of partnership opportunities.
Analyst forecasts reflect a highly optimistic outlook, echoing positive retail sentiment. The ongoing SPARTA trial also unveils compelling data, suggesting a more profound story from a long term perspective.
Is APLM set for a short squeeze akin to TPST? Time will tell…. Nonetheless, within this realm, APLM seems fundamentally undervalued, making it a stock to keep a close eye on as more developments arise.
We will update you on APLM when more details emerge, subscribe to Microcapdaily to follow along!
Subscribe to Our 100% Free Penny Stock Newsletter. We Have Something Big Coming!
Disclosure: We have not been compensated for this article/video. MicroCap Daily is not an investment advisor; this article/video does not provide investment advice. Always do your research, make your own investment decisions, or consult with your nearest financial advisor. This article/video is not a solicitation or recommendation to buy, sell, or hold securities. This article/video is our opinion, is meant for informational and educational purposes only, and does not provide investment advice. Past performance is not indicative of future performance.
BioPharma
Processa Pharmaceuticals (NASDAQ: PCSA): Revolutionizing Cancer Treatment through Next-Generation Chemotherapies
Published
8 months agoon
November 23, 2023
Processa Pharmaceuticals’ (NASDAQ: PCSA) shares skyrocketed by over 260% from their October lows of $0.18 per share. Shares peaked at $0.65 on November 14th and more recently has consolidated around $0.43. The reason for this surge was not immediately clear given the lack of material events. However upon digging deeper, we’ve uncovered a very interesting aspect driving the sudden rise. Let’s take a closer look at their pipeline and company history to understand what potential developments might be on the horizon and whether the current levels are justifiable.
 Background:
Background:
PCSA is a clinical stage Pharmaceutical company that’s working to create better versions of chemotherapy that’s safer and more effective. More specifically called, “Next Generation Chemotherapy” or NGCs for short – they’re essentially just modified versions of existing FDA-approved oncology drugs.
The objective is of course to improve clinical outcomes and have less adverse events (AEs). They do this through altering the metabolism and distribution of the drug while maintaining the main mechanism that kills the actual cancer cells.
It’s all based off over 30 years of drug development expertise. Although efficacy and safety can vary drastically depending on the type of chemotherapy, it’s widely known that undergoing chemo is undeniably challenging for patients due to the potential negative impact on their well-being.
PCSA has a few clinical drugs that are based on Capecitabine, Gemcitabine, and Irinotecan.
Respective problems that occur with these drug treatments:
- Low treatment response with high side effect profile.
- High drug resistance and/or acquired resistance; administered as IV
- Significant side-effect profile limits dosing and drug use.
PCSA is actively working to achieve:
- Actively minimizing Adverse Events (AEs), consequently broadening the patient base.
- Developing an oral therapy that accelerates the action of cancer-killing molecules while enhancing their quantity to mitigate drug resistance.
- Ensuring that cancer-killing molecules selectively enter cancer cells rather than normal cells, enhancing effectiveness and reducing toxicity.
Subscribe to Microcapdaily.com Right Now by entering your Email in the box below.
Subscribe to Our 100% Free Penny Stock Newsletter. We Have Something Big Coming!
What Happened:
We took a closer look at PCSA’s SEC filings and found something interesting: in October, there were a lot of Form 4 submissions. This seems to be the main reason for the sudden increase in PCSA’s valuation. Between October 10th and October 12th, insiders bought over 590,000 shares, and it wasn’t just one person—several insiders made these purchases. More often than not, this suggests a high expectation for something big happening soon.
Management Team:
Another important aspect to consider is the leadership team. It’s a fundamental step in conducting your due diligence since past achievements and expertise often pave the way for future success. Let’s swiftly overview just two key individuals in the company. For more information on others, click here.
George Ng | CEO:
Mr. Ng is a highly experienced figure in the life sciences industry with an extensive career involving founding and leading various companies. Notably, at Scilex Pharmaceuticals, Inc. (now, Scilex Holding Co (NASDAQ:SCLX)), where he played a key role in the company’s journey from development and clinical trials to NDA submission, FDA approval, a substantial $140 million financing round, the successful commercial launch of their first FDA-approved drug product, and the ultimate sale of the company. His career history includes a range of senior management positions across globally recognized biotechnology and pharmaceutical companies like Sorrento Therapeutics, Inc. (NASDAQ: SRNE), BioDelivery Sciences International, Inc. (NASDAQ: BDSI), Spectrum Pharmaceuticals, Inc. (NASDAQ: SPPI), and Alpharma, Inc. (NYSE:ALO), which is now part of Pfizer Inc. In these roles, Mr. Ng played pivotal roles in strategy, development, fundraising, and the commercialization of multiple pharmaceutical drug products.
David Young | President, Research & Development:
Dr. Young possesses extensive expertise in pharmaceutical research and development, spanning over three decades. As the Chief Scientific Officer of Questcor Pharmaceuticals from 2009 to 2014, he collaborated with the FDA to modernize Acthar Gel’s label and secure FDA approval for Infantile Spasms. His impactful involvement contributed significantly to Questcor’s transformation from near bankruptcy in 2007 to an esteemed orphan drug specialty company, reaching a valuation of around $5.6 billion by 2014. He was also the Founder and CEO of GloboMax LLC, a CRO specializing in FDA drug development that was purchased by ICON plc in 2003. Preceding his tenure at GloboMax, Dr. Young held a tenured Associate Professor position at the University of Maryland School of Pharmacy, leading a team of 30 individuals dedicated to evaluating the biological properties of drugs and drug delivery systems in both animals and humans.
Young’s experience extends even further: he served on FDA Advisory Committees, played a key role in an FDA-funded Clinical Pharmacology contract, oversaw the evaluation of oral products in a UMAB-FDA contract leading to FDA Guidance improvements, educated FDA reviewers for five years, participated in NIH grant review committees, and co-led a National Cancer Institute contract evaluating new oncology drugs.
Overall, Young’s met more than 100 times with the FDA on more than 50 drug products and played a vital role in over 30 NDA/supplemental NDA approvals. Additionally, he has over 150 published works, including presentations to the FDA, and numerous talks at scientific and investment gatherings.
It’s certainly significant that someone with such vast experience as Dr. Young is associated with PCSA, valued at ~10M at time of writing. Considering this executive’s extensive background in navigating FDA approvals, we hold a strong positive outlook on its potential trajectory. Dr. Young’s substantial insider purchases also strongly hint at positive developments ahead.
Subscribe to Microcapdaily.com Right Now by entering your Email in the box below.
Subscribe to Our 100% Free Penny Stock Newsletter. We Have Something Big Coming!
Phase 1B Trial Update:
The last Phase 1B trial update is written in a fairly scientific format, so we’ve taken the liberty of simplifying the release to better your understanding.
The Phase 1B trial of NGC-Cap in patients with gastrointestinal cancer aims to create a personalized treatment method that could be safer and more effective.
NGC-Cap is a treatment that mixes PCS6422, which stops an enzyme called DPD, with small amounts of a common chemotherapy drug, capecitabine. This chemotherapy drug turns into 5-FU in the body. DPD helps change 5-FU into another substance called FBAL, which usually causes strong side effects, making it tough to give higher doses of the chemotherapy treatment.
Processa discovered that regularly checking DPD levels, (shown by the substance FBAL), could help understand how each patient reacts to different NGC-Cap doses. This information might help doctors create specific treatment plans for each patient, aiming to reduce side effects and make the treatment work better while keeping the patient safe and comfortable.
Capecitabine is among the most widely used chemotherapy drugs and is dosed based on a standard dosage regimen for all patients. That said, many patients cannot tolerate that dose and must either have their dose reduced or have their treatment interrupted, which means ultimately means it’s less effective overall.
The discoveries made in this interim data is very promising and could mean that PCSA is now capable of personalized treatment plans, helping patients potentially handle the treatment much better without AEs.
Key Catalysts and Considerations:
There’s supposedly more updates soon for PCSA’s two main drugs, PCS6422/NGC-Cap and PCS3117/NGC-Gem.
For PCS6422/NGC-Cap, they’re finishing up the Phase 1B study and planning the Phase 2 trial based on feedback from the FDA.
With PCS3117/NGC-Gem, they’re working closely with the FDA to plan the drug’s development and how it will be tested.
There’s also been a recent change in leadership, bringing in a new CEO, George Ng in August 2023. This new leader has experience in turning companies around, developing business, and as mentioned, knows a lot about cancer treatment. The company is also considering licensing non-core assets to bring in money without dilution.
Overall PCSA’s new chemotherapy drugs look promising for treating a diverse range of cancers, meaning a potential billion-dollar US market opportunity alone. They have experienced leaders who know how to get medicines approved and make successful deals.
As of June 2023, they have about $8.7 million in cash, meaning a runway into the second half of 2024. They’re also thinking looking into potential partnership opportunities to further to develop their pipeline to avoid dilution.
Some of these events would be considered significant catalysts, potentially propelling the company to new heights. With a relatively low float, price fluctuations could be volatile in nature—leading to substantial gains or complete loss of investment. However at these levels, PCSA certainly seems like an interesting opportunity for potential upside, be sure to keep them on your radar.
We will update you on PCSA when more details emerge, subscribe to Microcapdaily to follow along!
Subscribe to Our 100% Free Penny Stock Newsletter. We Have Something Big Coming!
Disclosure: We have not been compensated for this article/video. MicroCap Daily is not an investment advisor; this article/video does not provide investment advice. Always do your research, make your own investment decisions, or consult with your nearest financial advisor. This article/video is not a solicitation or recommendation to buy, sell, or hold securities. This article/video is our opinion, is meant for informational and educational purposes only, and does not provide investment advice. Past performance is not indicative of future performance.
Recent Posts

Clean Vision Corp (OTC: CLNV): Overcoming the Plastic Waste Crisis

Meta Materials (NASDAQ: MMAT): More Due Diligence and Exploring Latest Developments

Integrated Cannabis Solutions’ (OTC: IGPK) 633% Surge: Exploring Catalysts, Company Overview, and Growth Potential in 2024

Sonoma Pharmaceuticals (NASDAQ: SNOA): Potential Surge to Speculations – What Lies Ahead?

1847 Holdings (NYSE: EFSH) Soars: Insights, Acquisitions, and What Lies Ahead
Trending
-

 Uncategorized2 years ago
Uncategorized2 years agoMeta Materials Inc (OTCMKTS: MMTLP) Enormous Short Position in Trouble as Next Bridge Hydrocarbons Set to Stop Trading (George Palikaras & John Brda on Corporate Action)
-

 Micro Cap Insider3 years ago
Micro Cap Insider3 years agoMedium (KOK PLAY) The Parabolic Rise of Metal Arts (OTCMKTS: MTRT)
-

 Media & Technology3 years ago
Media & Technology3 years agoHealthier Choices Management Corp. (OTCMKTS: HCMC) Powerful Comeback Brewing as PMI Patent Infringement Lawsuit Moves Forward
-
Media & Technology4 years ago
AMECA Mining RM; the Rise of Southcorp Capital, Inc. (OTCMKTS: STHC)
-

 Media & Technology3 years ago
Media & Technology3 years agoSNPW (Sun Pacific Holding Corp) Power Brewing: 50MW solar farm project in Durango Mexico MOU with Atlas Medrecycler 48,000 SF New Partnership Queensland Australia Solar Farm.
-

 BioPharma2 years ago
BioPharma2 years agoAsia Broadband (OTCMKTS: AABB) On the Move Northbound Since Sub $0.08 Dip as Crypto Innovator Elevates AABB Crypto Exchange & Enters the NFT Space
-

 Uncategorized2 years ago
Uncategorized2 years agoMeta Materials Inc (OTCMKTS: MMTLP) Short Squeeze S-1a4 Filing Signals S1 Approval Could Be Days Away (Next Bridge Hydrocarbons Spin-Off)
-

 BioPharma3 years ago
BioPharma3 years agoHumbl Inc (OTCMKTS: HMBL) Major Reversal as Powerful Advisor Rejoins the Team & Looks to Uplist to Major Exchange